by Jackie Dosal, MD
Is my sunscreen going to kill me? Perform a search on sunscreen safety and some scary stuff shows up! Retinyl palmitate causes skin cancer. Oxybenzone causes estrogen-like effects on the body. I have seen these statements repeated over and over again all over social media and in real life. I recently heard from a patient, “I was using Neutrogena but then stopped because I heard it wasn’t so good for you.†How do we protect ourselves and our families from skin cancer without any nasty side effects?
I’d like to offer up my take — from a organic-loving, Whole-Foods-shopping, yoga-practicing, green-tea-drinking MD (dermatologist), with an eye for interpreting both media and science…
For those not able to read the whole post (we are all busy, I know!), here’s the quick summary of my findings. You can read on for more info too!
Sunscreen won’t cause hormonal disruption — it would take 200 years of application to even reach questionable levels of exposure.
Sunscreen won’t cause skin cancer — the use of sunscreen is directly correlated to the prevention of skin cancer. Retinyl palmitate is an anti-oxidant that occurs naturally in the skin.
Nanosize sunscreens are safe for use, as they clump in real life, preventing them from being absorbed.
I’ve researched the issues of sunscreen safety personally, and WOW! It’s confusing and contradictory. I’ll at least say this— we always need to critically and scientifically analyze the sunscreen ingredients we use for both efficacy and safety. And just as important, let’s not perpetuate myths that are based on unsound science. If you are interested in sorting through the muddy waters of sunscreen information — read on!
Environmental Working Group’s Rankings of Sunscreens.
Most of the information about safety stems from the Environmental Working Group’s sunscreen rankings. The Environmental Working Group is a not-for-profit organization that searches to identify harmful chemicals in our environment. While I applaud the intentions of the EWG to find safe and environmentally responsible materials, we need to be fair and look at these issues seriously, and not just solely how presented by the EWG.
Do sunscreens cause hormonal disruption?
The EWG advises avoiding any products with oxybenzone (an excellent UVA and UVB blocker) as it may cause effects similar to estrogen. The concern comes from a study performed in rats, where the rats were fed supratherapeutic doses of sunscreen (let me say that again, the rats ATE sunscreen at megadoses), and the size of their uterus enlarged.
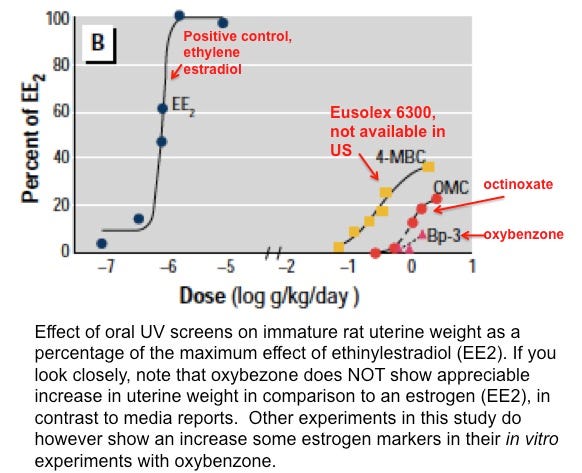
There is a monstrous difference between eating super high doses of sunscreen and applying it to your skin daily — and concluding that topical use in human sunscreen causes estrogenic effects is an unrealistic stretch. A study in 2011 in JAMA Dermatology showed that it would take 200 years of daily sunscreen application to reach the same amount exposure as the rats in this study. To the author’s credit, their word-for-word conclusion in the abstract was: “Our findings indicate that UV screens should be tested for endocrine activity…†The authors do not say that the UV screens cause hormone activity in humans… they can’t. In order to substantiate their EWG’s claim, you would need to show hormone disruption in real live humans.
Luckily, such a study was performed. Human patients applied more than 3 times the real-life quantity of a high percentage oxybenzone sunscreen (10% oxybenzone, compared to the commercially available 6% oxybenzone) to their whole body daily for one week. Oxybenzone was in fact detected in the urine of the volunteers (as have some other sunscreen agents). Sound the alarm bells! — this is concerning. It indicates that there is some absorption from topical use (medications that are applied directly to the surface of the skin). However, the body quickly excreted it through the urine (which is a good thing). Much can be said about the fact that it was absorbed, but the take-home message was that the researchers were UNABLE to show a hormone disruption despite this absorption (again, this was at 3 times real-life dosage). There was no accumulation of the ingredients over time.
So we have a study in humans of real-life sunscreen use that shows no hormonal effect, or a study where rats ate sunscreen — which do you believe?
To be fair, when researching this blog post, I did come across an alarming number of lab and animal studies showing absorption of various sunscreen agents, with some experiments suggesting hormonal disruption. There were almost as many that showed no absorption nor hormone disruption. A good summary of those studies can be found here. But we have to look at what’s important… no human studies have shown hormonal disruption, and that is what counts.
Of note, it is important to mention that oxybenzone can cause allergic skin reaction in a fair percentage of people, so there should be some caution with its use.
And by the way, soy is weakly estrogenic. Where are the cries for banning or avoiding soy milk because of hormonal activity? There are many natural skin care products on the market containing soy, several of which I recommend and are excellent. A little perspective is important, and we need to remember that topical use is very different from ingestion.
Let’s move on to sunscreen and skin cancer.
Does my sunscreen increase my risk of skin cancer?
Retinyl palmitate is an interchangeable form of retinol that is present naturally in our skin, and this should already be a clue that it’s not dangerous. Retinyl palmitate is not just in sunscreens; is approved by the FDA for use in prescription and over the counter medications, as well as a food additive in dairy products and cereals. It was selected for testing due to its prevalent use in common agents, not for any concerns of safety up to that point.
Once again, we have to look at the details of the study that created the buzz. It was never published in a peer-reviewed journal (the standard for medical literature — data needs to be vetted by people in-the-know), and was not performed in humans (also a huge red flag). It was performed in hairless mice, known to have an already increased risk of skin cancer, and these mice were burned with and without retinyl palmitate (RP). The studies showed the production of oxidative damage and increased risk of malignancy. However, these studies take RP out of human context, which is a very complex interactive antioxidant system. According to Dr. Steven Wang, the lead investigator in the study, “when a sunscreen with retinyl palmitate is applied to the skin, a number of antioxidants work together to alleviate the risk of free radical formation seen in these in vitro experiments. If studied on its own — outside of this environment — its antioxidant properties can rapidly be exhausted, allowing the production of oxygen radicals. In these non-human studies, retinyl palmitate was the only compound studied — making the biological relevance of these findings to humans unclear.†Even the lead investigator is not ready to recommend avoiding retinyl palmitate.
The final and probably most convincing evidence of the safety of retinyl palmitate comes from real life use in humans. We often use the family of retinoids (of which retinyl palmitate is a member), as a pill for the prevention of skin cancers in those most at risk (immunosuppressed organ transplant patients). We also have decades of experience using retinoids in acne medications; and topical retinoids are one of the best anti-aging creams on the market — I personally use one (Retin-A) every night. Undeniable human research shows it can reverse oxidative damage and signs of aging, keeping the skin healthy and youthful.
If you are interested in more, I encourage you to read this study by the Journal of the American Academy of Dermatology detailing the properties of retinyl palmitate and discussing its safety.
What are nano particles, and are they safe?
Nanoparticles are molecules that have been divided into nanometers (one billionth of a meter). By making the molecules small, it makes sunscreens more cosmetically acceptable and elegant, thus making it more attractive for people to use (i.e. not thick, sticky, or opaque like the old sunscreens found on lifeguards’ noses).
Concerns arose that these molecules might be small enough to penetrate through the skin into the bloodstream. Time and time again, human studies have shown that these particles are unable to penetrate through the skin because, in nature, these nanoparticles aggregate into clumps that are too large to penetrate the skin. There was also some thought that these ingredients could increase reactive oxidative species (ROS). We now know that this is meaningless if tested outside of the complex human antioxidative system. With this in mind, we should all embrace these new technologies that makes sunscreens less opaque, pastey, and more consumer friendly!

Nanoparticles clump on the skin. Photo credit: PMID: 22123418
Be Cautious of Media Sound Bites
The EWG extrapolates data (usually from animal studies, not humans), and uses scare tactics to make us feel like we are hurting our children if we use certain products. What I don’t like is that they ignore actual sound studies that show safety in humans. Their sound bites are alarming and catchy for headlines. I’ve actually seen website titles citing the EWG reading: “The Most Dangerous Sunscreens For Kids (And They’re Probably Hiding in Your Beach Bag).†Talk about mommy guilt!
I love this story which explains why we need to critically interpret how the media presents headlines: Chocolate Causes Weight Loss!
Background: A scientist wanted to show just how easy it was to convince the media to pick up a poorly designed faux scientific experiment that was catchy for headlines. It was a completely fake, but intriguing idea — eating chocolate can help loose weight. It turns out, it was really easy to get experienced journalists to pick up the headline! Read here: I Fooled Millions into Thinking Chocolate Helps Weight Loss. A critical eye is really needed in these days of rapid information sharing.
What does this all mean?
I don’t agree with the EWG rankings. Many of the poorly ranking sunscreens are very reputable and excellent brands. You can’t take the data from one animal or lab study and ignore other science and human experiments that are in disagreement. That’s not how science works. That’s the reason why thousands of drugs that work in animals never get to the market — because animal studies and in vitro studies often don’t translate into efficacy in the complex environment of the human body.
While in general, I’m in favor of the organic movement, real foods, and responsible farming practices, I often laugh when I see websites recommending chemical-free everything, including sunscreens. Chemicals are in everything, and there is no such thing as a purely chemical-free susncreen. Of course, live in moderation — extreme use of any susbtance can lead to some bad effects, but to say that normal day-to-day use of products causes the same effect seen with extreme use or oral ingestion doesn’t make any sense.
The dose is the poison, not the chemical itself. Almost any substance, including natural or beneficial substances, can be toxic under certain conditions, such as high temperatures, pH, concentrations, or when in the presence of other chemicals. Even too much Vitamin C can be lethal to skin cells at certain doses. The age-old mantra still holds true — everything in moderation.
Let’s thank the EWG for their laudable efforts in keeping us safe, but ask them not to demonize valuable products when the science shows otherwise. I completely agree that more testing needs to be done on household and cosmetic ingredients, but I wish they were a little more unbiased when it came to promotion of their agenda. And the media perpetuates these news snippets with negligible investigation into the validity of the EWG claims. It serves our society poorly.
Feel good about protecting your family — and apply that sunscreen this summer!










Recent Comments